Heart diseases, carcinoma, chronic pulmonary diseases, Alzheimer's disease, pneumonia and influenza are all linked with this enzyme that is produced by our body.1 This enzyme is called 5-lipoxygenase or 5-LOX for short, which, when generated by the body in excessive amounts, gives rise to a whole cascade of dangerous inflammatory reactions.
Ironically enough, it was this systemic inflammatory reaction caused by 5-lipoxygenase that protected our ancestors from infective diseases. Since these diseases are being increasingly eliminated, the anti-inflammatory reaction of 5-lipoxygenase has not only become unnecessary, but also deadly.2-5 Scientists have been familiar with this enzyme for some time due to its role in occurrence of arthritis, asthma and Crohn’s disease, which is why the pharmaceutical industry aims to develop a safe drug to alleviate the effect of 5-lipoxygenase. However, the currently available “5-LOX inhibitors” are not safe enough to be taken without medical supervision.6,7 Luckily, there are various foodstuffs and plants that effectively prevent the effect of 5-lipoxygenase.
How does a 5-lipoxygenase surplus have a detrimental effect on the health?
As a result of ageing and/or bad diet, our body generates excessive amounts of arachidonic acid. In order to neutralise arachidonic acid, increased amounts of 5-lipoxygenase (5-LOX) are produced in our system. Increased 5-LOX enzyme values and end products of arachidonic acid breakdown are closely connected with middle and old age diseases.
The diagram below clearly shows how typical Western diet food causes a surplus of arachidonic acid to be created, resulting in harmful consequences of increased 5-lipoxygenase values.
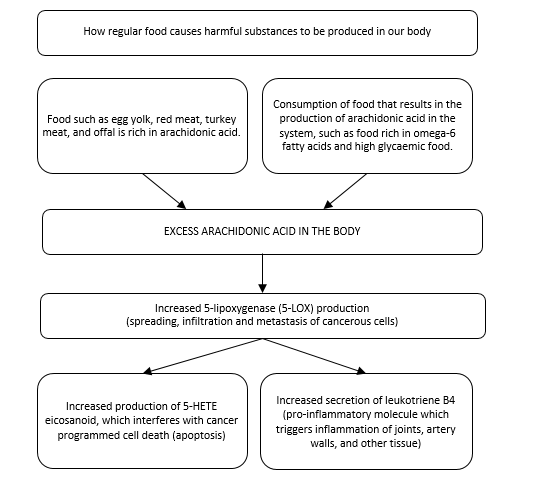
The diagram depicts how the body metabolises Western diet via 5-lipoxygenase
"5-lipoxygenase stimulates the production of pro-inflammatory molecules in the body called leukotrienes.8 Hundreds of published scientific studies have established a link between leukotrienes and the occurrence of cardiovascular disease9, carcinoma10-16, arthritis17-19, and respiratory diseases, such as asthma and chronic obstructive pulmonary disease (COPD)8. These molecules are linked with Alzheimer's disease20-25, inflammatory bowel diseases 26-28 and osteoporosis.29,30"
By switching to healthier dietary patterns, the 5-lipoxygenase level decreases, even though optimal suppression of 5-LOX and its harmful effects paired with typical Western nutrition can pose quite a challenge. Fortunately, frankincense extract has proved an effective inhibitor against this enzyme.
How to prevent excessive 5-lipoxygenase production For millennia, ancient cultures acknowledged frankincense and myrrh resin both as a source of pleasant fragrances and as medicine against rheumatism, herpes, and gastrointestinal disorders. In modern studies, the medicinal value of frankincense was confirmed with the discovery of boswellic acids contained within this resin. The 3-acetyl-11-keto-beta-boswellic acid (AKBA) has turned out to be the most effective in inhibiting 5-lipoxygenase and preventing the creation of pro-inflammatory leukotrienes.39,40 It has been confirmed through animal experiments that frankincense extracts possess a strong anti-inflammatory effect, especially for mitigating joint inflammation in arthritis.41 Initial human studies have yielded similar feasible results, thus confirming the traditional use of frankincense. Frankincense extracts have proved effective in patients suffering from rheumatoid arthritis, Crohn’s disease, ulcerative colitis and asthma.39,42 Throughout the millennia of frankincense use, no significant side-effects have been observed. It has been corroborated by recent toxicology and safety studies that frankincense extracts have a wide safety range, both in external and internal application.43
How to prevent excessive 5-lipoxygenase production
For millennia, ancient cultures acknowledged frankincense and myrrh resin both as a source of pleasant fragrances and as medicine against rheumatism, herpes, and gastrointestinal disorders.
In modern studies, the medicinal value of frankincense was confirmed with the discovery of boswellic acids contained within this resin. The 3-acetyl-11-keto-beta-boswellic acid (AKBA) has turned out to be the most effective in inhibiting 5-lipoxygenase and preventing the creation of pro-inflammatory leukotrienes.39,40 It has been confirmed through animal experiments that frankincense extracts possess a strong anti-inflammatory effect, especially for mitigating joint inflammation in arthritis.41
Initial human studies have yielded similar feasible results, thus confirming the traditional use of frankincense. Frankincense extracts have proved effective in patients suffering from rheumatoid arthritis, Crohn’s disease, ulcerative colitis and asthma.39,42 Throughout the millennia of frankincense use, no significant side-effects have been observed. It has been corroborated by recent toxicology and safety studies that frankincense extracts have a wide safety range, both in external and internal application.43
Renarthro – a synergy formula of frankincense and myrrh extracts plus colostrum
 The Croatian company Farmavita has developed Renarthro (www.renarthro.com), a product which contains frankincense extract, myrrh extract and colostrum. It has been found in laboratory studies that myrrh extract prevents the effect of cartilage-degrading enzymes (hyaluronidase, collagenase). It has also been confirmed that the addition of myrrh extract enhances the effect of other plants 20-fold. Other laboratory studies have shown that PRP peptides found in milk improve the effect of other ingredients by several times. The patent-protected innovation provided by Farmavita is the fact that the combination of these three ingredients forms a unique synergy that is several times more effective than the individual components.
The Croatian company Farmavita has developed Renarthro (www.renarthro.com), a product which contains frankincense extract, myrrh extract and colostrum. It has been found in laboratory studies that myrrh extract prevents the effect of cartilage-degrading enzymes (hyaluronidase, collagenase). It has also been confirmed that the addition of myrrh extract enhances the effect of other plants 20-fold. Other laboratory studies have shown that PRP peptides found in milk improve the effect of other ingredients by several times. The patent-protected innovation provided by Farmavita is the fact that the combination of these three ingredients forms a unique synergy that is several times more effective than the individual components.
The role of inflammation processes in our body following the occurrence of atherosclerosis (blood vessel thickening and congestion) has been known for over 20 years. In 1991, it was discovered that one of atherosclerosis factors is a molecule that is the end product of a process beginning with the effect of 5-lipoxygenase.50,51 Thus, for more than 10 years, there have been attempts to isolate 5-LOX inhibitors that would prevent or even reverse the ageing of the cardiovascular system.52 Frankincense extracts have turned out to be one of the best natural 5-LOX inhibitors when it comes to atherosclerosis occurrence.53
Protection of heart and blood vessels
Frankincense extracts have multiple complementary effects in vascular tissue. They strongly affect free radical binding.54 Also, they decrease total cholesterol by up to 48% and increase “good cholesterol” (high-density lipoprotein cholesterol – HDL) by up to 30%.55 However, it is the effect of frankincense extract on 5-lipoxygenase inhibition and on other inflammatory factors that interests cardiologists the most. In the human capillary endothelium, frankincense extracts have a significant effect on the prevention of damage to blood vessel walls connected to atherosclerosis43,56. It is estimated that 113 genes involved in the occurrence of vascular inflammation are positively modulated by frankincense.56,57
Extracts rich in AKBA, the main bioactive agent in frankincense, have shown a 50% reduction in atherosclerosis-induced damage.58 The activity of several different blood coagulation factors is also substantially reduced. A similar effect is attributed to guggulsterones found in myrrh extract.54
Neuroprotection and brain health
It was discovered that a constituent of frankincense is a potential stimulator of the brain receptor system called TRPV3, which is responsible for regulating anxiety and has an effect similar to antidepressants.59
One of the main components of frankincense obtained from Boswellia carteri trees, called incensole acetate, inhibits inflammation mediators in cerebral tissue – an effect which gives significant hope in treatment of cerebral tissue damage.60 Dangerous inflammatory cerebral reactions can inflict damage on cerebral tissue in a matter of a few hours, causing long-term neurological deficiencies.
Incensole acetate particularly acts as an inhibitor of cerebral cell degeneration in the hippocampus, the region of brain responsible for memory.60This effect has been confirmed by animal studies. Laboratory rats that were given a memory loss inducing substance, paired with frankincense extract, yielded significantly better results in spatial orientation and learning tests compared to animals that were not given frankincense extract.61 Similar results were obtained with animals that were not given the memory loss inducing substance. Extracts obtained from Boswellia papyrifera, one of 5 Boswellia species used in the production of fragrant frankincense resin, were tested on rats and yielded animals with better spatial orientation, allowing them to find their way through a labyrinth quicker.62
Carcinoma protection
The effect of the boswellic acid, one of the main frankincense components, is achieved through several mechanisms which prevent the proliferation (spreading) of cancerous cells. AKBA induces apoptosis, i.e. programmed cell death, by affecting the so-called “death receptor” on cell surface and causing “suicide” of the carcinoma cell.63
Boswellic acid also blocks intercellular communication between carcinoma cells responsible for cellular replication.64 AKBA also impedes tumour growth by inhibiting the vascular endothelial growth factor (VEGF) used by cells for development of necessary new blood vessels.65
Finally, it was recently discovered that AKBA can be effective when inhibiting the cellular receptor CXCR4, used by carcinoma cells to induce metastases and considered the cause of death by cancer in 90% of cases. To this day, frankincense extracts have also shown promising results when it comes to reducing the growth of malignant cells, carcinoma replication, or metastases of prostate, bladder, uterus cervix, bowels, as well as multiple myeloma and bone marrow cancer.66-7
Frankincense extracts alleviate arthritis pain
Other ways to mitigate the effect of 5-LOX enzyme
Typical Western diet has an abundance of omega-6 fatty acids, saturated fatty acids, refined carbohydrates, and arachidonic acid. Excessive consumption of such food and insufficient intake of food rich in omega-3 fatty acids significantly contributes to the development of systemic chronic inflammatory diseases.
Those who are mindful of their health use nutrients such as fish oil or turmeric which decrease the activity of 5-lipoxygenase in the system.109-111 Their effect on 5-LOX suppression also serves as a partial explanation for a number of benefits provided by these nutrients.
However, as people age, excessive 5-LOX production calls for more effective suppression methods. Herbal preparations with frankincense extract, coupled with healthier dietary habits, offer an effective natural method of selective 5-LOX inhibition.112,113
Conclusion
Excess 5-lipoxygenase causes the production of pro-inflammatory molecules, including leukotrienes. Inflammatory processes within the body, induced by 5-lipoxygenase, are linked with seven out of ten most common causes of death in the USA, including cancer and heart diseases.
Presently, there exists a small number of safe medical products that inhibit 5-LOX; however, they must be taken under a physician’s supervision. Fortunately, natural 5-LOX inhibitors that effectively inhibit the activity of this enzyme have been recently discovered. As evidenced by a large body of convincing information, frankincense components block inflammatory reactions caused by 5-LOX, the enzyme which accelerates the occurrence of numerous fatal ageing-induced diseases.
"All those who wish to prevent the harmful effect of this enzyme and prolong their healthy life should adopt an anti-inflammatory diet with reduced or eliminated use of red meat, offal, sausages, sugar, and high glycaemic food.When selecting dietary fats and oils, the ones with a high content of omega-3 unsaturated fats (olive oil, coconut oil, linseed oil, pumpkin seed oil, and fish oil) should be preferred.We also recommend one capsule of Renarthro daily, as a supplement to the anti-inflammatory diet."
Reference
1. http://www.cdc.gov/nchs/fastats/lcod.htm.
2. Chinnici CM, Yao Y, Pratico D. The 5-lipoxygenase enzymatic pathway in the mouse brain: young versus old. Neurobiol Aging. 2007 Sep;28(9):1457-62.
3. Chu J, Pratico D. The 5-lipoxygenase as a common pathway for pathological brain and vascular aging. Cardiovasc Psychiatry Neurol. 2009;2009:174657.
4. Graham FD, Erlemann KR, Gravel S, Rokach J, Powell WS. Oxidative stress-induced changes in pyridine nucleotides and chemoattractant 5-lipoxygenase products in aging neutrophils. Free Radic Biol Med. 2009 Jul 1;47(1):62-71.
5. Zou Y, Kim DH, Jung KJ, et al. Lysophosphatidylcholine enhances oxidative stress via the 5-lipoxygenase pathway in rat aorta during aging. Rejuvenation Res. 2009 Feb;12(1):15-24.
6. Abe M, Yoshimoto T. Leukotriene-lipoxygenase pathway and drug discovery. Nihon Yakurigaku Zasshi. 2004 Dec;124(6):415-25.
7. Chohnabayashi N, Sugiura R, Nishimura N. Adverse effects of leukotriene-antagonists. Nihon Rinsho. 2007 Oct 28;65 Suppl 8:272-6.
8. Sampson AP. FLAP inhibitors for the treatment of inflammatory diseases. Curr Opin Investig Drugs. 2009 Nov;10(11):1163-72.
9. Jawien J, Korbut R. The current view on the role of leukotrienes in atherogenesis. J Physiol Pharmacol. 2010 Dec;61(6):647-50.
10. Goodman LA, Jarrett CL, Krunkosky TM, et al. 5-Lipoxygenase expression in benign and malignant canine prostate tissues. Vet Comp Oncol. 2011 Jun;9(2):149-57.
11. Angelucci A, Garofalo S, Speca S, et al. Arachidonic acid modulates the crosstalk between prostate carcinoma and bone stromal cells. Endocr Relat Cancer. 2008 Mar;15(1):91-100.
12. Faronato M, Muzzonigro G, Milanese G, et al. Increased expression of 5-lipoxygenase is common in clear cell renal cell carcinoma. Histol Histopathol. 2007 Oct;22(10):1109-18.
13. Grant GE, Rubino S, Gravel S, et al. Enhanced formation of 5-oxo-6,8,11,14-eicosatetraenoic acid by cancer cells in response to oxidative stress, docosahexaenoic acid and neutrophil-derived 5-hydroxy-6,8,11,14-eicosatetraenoic acid. Carcinogenesis. 2011 Jun;32(6):822-8.
14. Kim EY, Seo JM, Cho KJ, Kim JH. Ras-induced invasion and metastasis are regulated by a leukotriene B4 receptor BLT2-linked pathway. Oncogene. 2010 Feb 25;29(8):1167-78.
15. Radmark O, Samuelsson B. Microsomal prostaglandin E synthase-1 and 5-lipoxygenase: potential drug targets in cancer. J Intern Med. 2010 Jul;268(1):5-14.
16. Ye YN, Liu ES, Shin VY, Wu WK, Cho CH. Contributory role of 5-lipoxygenase and its association with angiogenesis in the promotion of inflammation-associated colonic tumorigenesis by cigarette smoking. Toxicology. 2004 Oct 15;203(1-3):179-88.
17. Chen M, Lam BK, Luster AD, et al. Joint tissues amplify inflammation and alter their invasive behavior via leukotriene B4 in experimental inflammatory arthritis. J Immunol. 2010 Nov 1;185(9):5503-11.
18. Chen SH, Fahmi H, Shi Q, Benderdour M. Regulation of microsomal prostaglandin E2 synthase-1 and 5-lipoxygenase-activating protein/5-lipoxygenase by 4-hydroxynonenal in human osteoarthritic chondrocytes. Arthritis Res Ther. 2010;12(1):R21.
19. Mathis SP, Jala VR, Lee DM, Haribabu B. Nonredundant roles for leukotriene B4 receptors BLT1 and BLT2 in inflammatory arthritis. J Immunol. 2010 Sep 1;185(5):3049-56.
20. Chu J, Pratico D. 5-lipoxygenase as an endogenous modulator of amyloid beta formation in vivo. Ann Neurol. 2011 Jan;69(1):34-46.
21. Chu J, Pratico D. Pharmacologic blockade of 5-lipoxygenase improves the amyloidotic phenotype of an Alzheimer's disease transgenic mouse model involvement of gamma-secretase. Am J Pathol. 2011 Apr;178(4):1762-9.
22. Firuzi O, Zhuo J, Chinnici CM, Wisniewski T, Pratico D. 5-Lipoxygenase gene disruption reduces amyloid-beta pathology in a mouse model of Alzheimer's disease. FASEB J. 2008 Apr;22(4):1169-78.
23. Ikonomovic MD, Abrahamson EE, Uz T, Manev H, Dekosky ST. Increased 5-lipoxygenase immunoreactivity in the hippocampus of patients with Alzheimer's disease. J Histochem Cytochem. 2008 Dec;56(12):1065-73.
24. Klegeris A, McGeer PL. Cyclooxygenase and 5-lipoxygenase inhibitors protect against mononuclear phagocyte neurotoxicity. Neurobiol Aging. 2002 Sep-Oct;23(5):787-94.
25. Qu T, Manev R, Manev H. 5-Lipoxygenase (5-LOX) promoter polymorphism in patients with early-onset and late-onset Alzheimer's disease. J Neuropsychiatry Clin Neurosci. 2001 Spring;13(2):304-5.
26. Cuzzocrea S, Rossi A, Mazzon E, et al. 5-Lipoxygenase modulates colitis through the regulation of adhesion molecule expression and neutrophil migration. Lab Invest. 2005 Jun;85(6):808-22.
27. Mazzon E, Sautebin L, Caputi AP, Cuzzocrea S. 5-lipoxygenase modulates the alteration of paracellular barrier function in mice ileum during experimental colitis. Shock. 2006 Apr;25(4):377-83.
28. Rask-Madsen J, Bukhave K, Laursen LS, Lauritsen K. 5-Lipoxygenase inhibitors for the treatment of inflammatory bowel disease. Agents Actions. 1992;Spec No:C37-46.
29. Boyce BF, Hughes DE, Wright KR, Xing L, Dai A. Recent advances in bone biology provide insight into the pathogenesis of bone diseases. Lab Invest. 1999 Feb;79(2):83-94.
30. Mundy GR. Cytokines and growth factors in the regulation of bone remodeling. J Bone Miner Res. 1993 Dec;8 Suppl 2:S505-10.
31. Bell RL, Harris RR, Malo PE, et al. ABT-761 attenuates bronchoconstriction and pulmonary inflammation in rodents. J Pharmacol Exp Ther. 1997 Mar;280(3):1366-73.
32. Cuzzocrea S, Rossi A, Serraino I, et al. 5-Lipoxygenase knockout mice exhibit a resistance to pleurisy and lung injury caused by carrageenan. J Leukoc Biol. 2003 Jun;73(6):739-46.
33. Doi K, Hamasaki Y, Noiri E, et al. Role of leukotriene B4 in accelerated hyperlipidaemic renal injury. Nephrology (Carlton). 2011 Mar;16(3):304-9.
34. Gubitosi-Klug RA, Talahalli R, Du Y, Nadler JL, Kern TS. 5-Lipoxygenase, but not 12/15-lipoxygenase, contributes to degeneration of retinal capillaries in a mouse model of diabetic retinopathy. Diabetes. 2008 May;57(5):1387-93.
35. Kim DC, Hsu FI, Barrett NA, et al. Cysteinyl leukotrienes regulate Th2 cell-dependent pulmonary inflammation. J Immunol. 2006 Apr 1;176(7):4440-8.
36. Martinez-Clemente M, Claria J, Titos E. The 5-lipoxygenase/leukotriene pathway in obesity, insulin resistance, and fatty liver disease. Curr Opin Clin Nutr Metab Care. 2011 Jul;14(4):347-53.
37. Pace E, Profita M, Melis M, et al. LTB4 is present in exudative pleural effusions and contributes actively to neutrophil recruitment in the inflamed pleural space. Clin Exp Immunol. 2004 Mar;135(3):519-27.
38. Zhou YJ, Wang JH, Li L, Yang HW, Wen de L, He QC. Expanding expression of the 5-lipoxygenase/leukotriene B4 pathway in atherosclerotic lesions of diabetic patients promotes plaque instability. Biochem Biophys Res Commun. 2007 Nov 9;363(1):30-6.
39. Ammon HP. Boswellic acids (components of frankincense) as the active principle in treatment of chronic inflammatory diseases. Wien Med Wochenschr. 2002;152(15-16):373-8.
40. Sailer ER, Subramanian LR, Rall B, Hoernlein RF, Ammon HP, Safayhi H. Acetyl-11-keto-beta-boswellic acid (AKBA): structure requirements for binding and 5-lipoxygenase inhibitory activity. Br J Pharmacol. 1996 Feb;117(4):615-8.
41. Fan AY, Lao L, Zhang RX, et al. Effects of an acetone extract of Boswellia carterii Birdw. (Burseraceae) gum resin on adjuvant-induced arthritis in lewis rats. J Ethnopharmacol. 2005 Oct 3;101(1-3):104-9.
42. Ammon HP. Boswellic acids in chronic inflammatory diseases. Planta Med. 2006 Oct;72(12):1100-16.
43. Lalithakumari K, Krishnaraju AV, Sengupta K, Subbaraju GV, Chatterjee A. Safety and Toxicological Evaluation of a Novel, Standardized 3-O-Acetyl-11-keto-beta-Boswellic Acid (AKBA)-Enriched Boswellia serrata Extract (5-Loxin(R)). Toxicol Mech Methods. 2006;16(4):199-226.
44. http://www.cdc.gov/nchs/fastats/arthrits.htm.
45. Barnes DE, Yaffe K. The projected effect of risk factor reduction on Alzheimer's disease prevalence. Lancet Neurol. 2011 Jul 18.
46. http://www.cdc.gov/nchs/fastats/asthma.htm.
47. http://www.cdc.gov/nchs/fastats/cancer.htm.
48. http://www.cdc.gov/nchs/fastats/heart.htm.
49.http://www.cdc.gov/nchs/fastats/kidbladd.htm.
50. Hagihara H, Nomoto A, Mutoh S, Yamaguchi I, Ono T. Role of inflammatory responses in initiation of atherosclerosis: effects of anti-inflammatory drugs on cuff-induced leukocyte accumulation and intimal thickening of rabbit carotid artery. Atherosclerosis. 1991 Nov;91(1-2):107-16.
51. Hatmi M, Samama MM, Elalamy I. Prevention of thrombosis and vascular inflammation: importance of combined cyclooxygenase and 5-lipoxygenase inhibitors. J Mal Vasc. 2006 Feb;31(1):4-9.
52. Zweifel BS, Hardy MM, Anderson GD, Dufield DR, Pufahl RA, Masferrer JL. A rat air pouch model for evaluating the efficacy and selectivity of 5-lipoxygenase inhibitors. Eur J Pharmacol. 2008 Apr 14;584(1):166-74.
53. Tripathi YB, Reddy MM, Pandey RS, et al. Anti-inflammatory properties of BHUx, a polyherbal formulation to prevent atherosclerosis. Inflammopharmacology. 2004;12(2):131-52.
54. Kokkiripati PK, Bhakshu LM, Marri S, et al. Gum resin of Boswellia serrata inhibited human monocytic (THP-1) cell activation and platelet aggregation. J Ethnopharmacol. 2011 Jul 8.
55. Pandey RS, Singh BK, Tripathi YB. Extract of gum resins of Boswellia serrata L. inhibits lipopolysaccharide induced nitric oxide production in rat macrophages along with hypolipidemic property. Indian J Exp Biol. 2005 Jun;43(6):509-16.
56. Roy S, Khanna S, Shah H, et al. Human genome screen to identify the genetic basis of the anti-inflammatory effects of Boswellia in microvascular endothelial cells. DNA Cell Biol. 2005 Apr;24(4):244-55.
57. Roy S, Khanna S, Krishnaraju AV, et al. Regulation of vascular responses to inflammation: inducible matrix metalloproteinase-3 expression in human microvascular endothelial cells is sensitive to antiinflammatory Boswellia. Antioxid Redox Signal. 2006 Mar-Apr;8(3-4):653-60.
58. Cuaz-Perolin C, Billiet L, Bauge E, et al. Antiinflammatory and antiatherogenic effects of the NF-kappaB inhibitor acetyl-11-keto-beta-boswellic acid in LPS-challenged ApoE-/- mice. Arterioscler Thromb Vasc Biol. 2008 Feb;28(2):272-7.
59. Moussaieff A, Rimmerman N, Bregman T, et al. Incensole acetate, an incense component, elicits psychoactivity by activating TRPV3 channels in the brain. FASEB J. 2008 Aug;22(8):3024-34.
60. Moussaieff A, Shein NA, Tsenter J, et al. Incensole acetate: a novel neuroprotective agent isolated from Boswellia carterii.
J Cereb Blood Flow Metab. 2008 Jul;28(7):1341-52.
61. Hosseini M, Hadjzadeh MA, Derakhshan M, et al. The beneficial effects of olibanum on memory deficit induced by hypothyroidism in adult rats tested in Morris water maze. Arch Pharm Res. 2010 Mar;33(3):463-8.
62. Mahmoudi A, Hosseini-Sharifabad A, Monsef-Esfahani HR, et al. Evaluation of systemic administration of Boswellia papyrifera extracts on spatial memory retention in male rats.
J Nat Med. 2011 Jul;65(3-4):519-25.
63. Lu M, Xia L, Hua H, Jing Y. Acetyl-keto-beta-boswellic acid induces apoptosis through a death receptor 5-mediated pathway in prostate cancer cells. Cancer Res. 2008 Feb 15;68(4):1180-6.
64. Kunnumakkara AB, Nair AS, Sung B, Pandey MK, Aggarwal BB. Boswellic acid blocks signal transducers and activators of transcription 3 signaling, proliferation, and survival of multiple myeloma via the protein tyrosine phosphatase SHP-1. Mol Cancer Res. 2009 Jan;7(1):118-28.
65. Pang X, Yi Z, Zhang X, et al. Acetyl-11-keto-beta-boswellic acid inhibits prostate tumor growth by suppressing vascular endothelial growth factor receptor 2-mediated angiogenesis. Cancer Res. 2009 Jul 15;69(14):5893-900.
66. Frank MB, Yang Q, Osban J, et al. Frankincense oil derived from Boswellia carteri induces tumor cell specific cytotoxicity. BMC Complement Altern Med. 2009;9:6.
67. Park B, Sung B, Yadav VR, Cho SG, Liu M, Aggarwal BB. Acetyl-11-keto-beta-boswellic acid suppresses invasion of pancreatic cancer cells through the downregulation of CXCR4 chemokine receptor expression. Int J Cancer. 2011 Jul 1;129(1):23-33.
68. Yuan HQ, Kong F, Wang XL, Young CY, Hu XY, Lou HX. Inhibitory effect of acetyl-11-keto-beta-boswellic acid on androgen receptor by interference of Sp1 binding activity in prostate cancer cells. Biochem Pharmacol. 2008 Jun 1;75(11):2112-21.
69. Yadav VR, Prasad S, Sung B, et al. Boswellic acid inhibits growth and metastasis of human colorectal cancer in orthotopic mouse model by downregulating inflammatory, proliferative, invasive, and angiogenic biomarkers. Int J Cancer. 2011 Jun 23.
70. Liu JJ, Duan RD. LY294002 enhances boswellic acid-induced apoptosis in colon cancer cells. Anticancer Res. 2009 Aug;29(8):2987-91.
71. Bhushan S, Malik F, Kumar A, et al. Activation of p53/p21/PUMA alliance and disruption of PI-3/Akt in multimodal targeting of apoptotic signaling cascades in cervical cancer cells by a pentacyclic triterpenediol from Boswellia serrata. Mol Carcinog. 2009 Dec;48(12):1093-108.
72. Dreikorn K. The role of phytotherapy in treating lower urinary tract symptoms and benign prostatic hyperplasia. World J Urol. 2002 Apr;19(6):426-35.
73. Paubert-Braquet M, Cave A, Hocquemiller R, et al. Effect of Pygeum africanum extract on A23187-stimulated production of lipoxygenase metabolites from human polymorphonuclear cells. J Lipid Mediat Cell Signal. 1994 May;9(3):285-90.
74. Paubert-Braquet M, Mencia Huerta JM, Cousse H, Braquet P. Effect of the lipidic lipidosterolic extract of Serenoa repens (Permixon) on the ionophore A23187-stimulated production of leukotriene B4 (LTB4) from human polymorphonuclear neutrophils. Prostaglandins Leukot Essent Fatty Acids. 1997 Sep;57(3):299-304.
75. Safarinejad MR. Urtica dioica for treatment of benign prostatic hyperplasia: a prospective, randomized, double-blind, placebo-controlled, crossover study. J Herb Pharmacother. 2005;5(4):1-11.
76. Lopatkin NA, Sivkov AV, Medvedev AA, et al. Combined extract of Sabal palm and nettle in the treatment of patients with lower urinary tract symptoms in double blind, placebo-controlled trial. Urologiia. 2006 Mar-Apr (2):12, 14-9.
77. Mantovani F. Serenoa repens in benign prostatic hypertrophy: analysis of 2 Italian studies. Minerva Urol Nefrol. 2010 Dec;62(4):335-40.
78. Pavone C, Abbadessa D, Tarantino ML, et al. Associating Serenoa repens, Urtica dioica and Pinus pinaster. Safety and efficacy in the treatment of lower urinary tract symptoms. Prospective study on 320 patients. Urologia. 2010 Jan-Mar;77(1):43-51.
79. Quiles MT, Arbos MA, Fraga A, de Torres IM, Reventos J, Morote J. Antiproliferative and apoptotic effects of the herbal agent Pygeum africanum on cultured prostate stromal cells from patients with benign prostatic hyperplasia (BPH). Prostate. 2010 Jul 1;70(10):1044-53.
80. Ejike CE, Ezeanyika LU. Inhibition of the Experimental Induction of Benign Prostatic Hyperplasia: A Possible Role for Fluted Pumpkin (Telfairia occidentalis Hook f.) Seeds. Urol Int. 2011 Jun 28.
81. Schleich S, Papaioannou M, Baniahmad A, Matusch R. Extracts from Pygeum africanum and other ethnobotanical species with antiandrogenic activity. Planta Med. 2006 Jul;72(9):807-13.
82. Papaioannou M, Schleich S, Roell D, et al. NBBS isolated from Pygeum africanum bark exhibits androgen antagonistic activity, inhibits AR nuclear translocation and prostate cancer cell growth. Invest New Drugs. 2010 Dec;28(6):729-43.
83. Kulkarni RR, Patki PS, Jog VP, Gandage SG, Patwardhan B. Treatment of osteoarthritis with a herbomineral formulation: a double-blind, placebo-controlled, cross-over study. J Ethnopharmacol. 1991 May-Jun;33(1-2):91-5.
84. Chopra A, Lavin P, Patwardhan B, Chitre D. A 32-week randomized, placebo-controlled clinical evaluation of RA-11, an Ayurvedic drug, on osteoarthritis of the knees. J Clin Rheumatol. 2004 Oct;10(5):236-45.
85. Kimmatkar N, Thawani V, Hingorani L, Khiyani R. Efficacy and tolerability of Boswellia serrata extract in treatment of osteoarthritis of knee--a randomized double blind placebo controlled trial. Phytomedicine. 2003 Jan;10(1):3-7.
86. Sengupta K, Alluri KV, Satish AR, et al. A double blind, randomized, placebo controlled study of the efficacy and safety of 5-Loxin for treatment of osteoarthritis of the knee. Arthritis Res Ther. 2008;10(4):R85.
87. Marone PA, Lau FC, Gupta RC, Bagchi M, Bagchi D. Safety and toxicological evaluation of undenatured type II collagen. Toxicol Mech Methods. 2010 May;20(4):175-89.
88. Bagchi D, Misner B, Bagchi M, et al. Effects of orally administered undenatured type II collagen against arthritic inflammatory diseases: a mechanistic exploration. Int J Clin Pharmacol Res. 2002;22(3-4):101-10.
89. Deparle LA, Gupta RC, Canerdy TD, et al. Efficacy and safety of glycosylated undenatured type-II collagen (UC-II) in therapy of arthritic dogs. J Vet Pharmacol Ther. 2005 Aug;28(4):385-90.
90. D'Altilio M, Peal A, Alvey M, et al. Therapeutic Efficacy and Safety of Undenatured Type II Collagen Singly or in Combination with Glucosamine and Chondroitin in Arthritic Dogs. Toxicol Mech Methods. 2007;17(4):189-96.
91. Trentham DE, Dynesius-Trentham RA, Orav EJ, et al. Effects of oral administration of type II collagen on rheumatoid arthritis. Science. 1993 Sep 24;261(5129):1727-30.
92. Crowley DC, Lau FC, Sharma P, et al. Safety and efficacy of undenatured type II collagen in the treatment of osteoarthritis of the knee: a clinical trial. Int J Med Sci. 2009;6(6):312-21.
93. Hunt CD. Regulation of enzymatic activity: one possible role of dietary boron in higher animals and humans. Biol Trace Elem Res. 1998 Winter;66(1-3):205-25.
94. Scorei IR. Calcium fructoborate: plant-based dietary boron as potential medicine for cancer therapy. Front Biosci (Schol Ed). 2011;3:205-15.
95. Scorei R, Cimpoiasu VM, Iordachescu D. In vitro evaluation of the antioxidant activity of calcium fructoborate. Biol Trace Elem Res. 2005 Nov;107(2):127-34.
96. Scorei R, Ciubar R, Iancu C, Mitran V, Cimpean A, Iordachescu D. In vitro effects of calcium fructoborate on fMLP-stimulated human neutrophil granulocytes. Biol Trace Elem Res. 2007 Jul;118(1):27-37.
97. Scorei R, Mitrut P, Petrisor I, Scorei I. A Double-Blind, Placebo-Controlled Pilot Study to Evaluate the Effect of Calcium Fructoborate on Systemic Inflammation and Dyslipidemia Markers for Middle-Aged People with Primary Osteoarthritis. Biol Trace Elem Res. 2011 May 24.
98. Scorei RI, Ciofrangeanu C, Ion R, et al. In vitro effects of calcium fructoborate upon production of inflammatory mediators by LPS-stimulated RAW 264.7 macrophages. Biol Trace Elem Res. 2010 Jun;135(1-3):334-44.
99. Scorei RI, Rotaru P. Calcium Fructoborate-Potential Anti-inflammatory Agent. Biol Trace Elem Res. 2011 Jan 28.
100. Wagner CC, Ferraresi Curotto V, Pis Diez R, Baran EJ. Experimental and theoretical studies of calcium fructoborate. Biol Trace Elem Res. 2008 Apr;122(1):64-72.
101. Ali AA, Lewis SM, Badgley HL, Allaben WT, Leakey JE. Oral glucosamine increases expression of transforming growth factor beta1 (TGFbeta1) and connective tissue growth factor (CTGF) mRNA in rat cartilage and kidney: implications for human efficacy and toxicity. Arch Biochem Biophys. 2011 Jun 1;510(1):11-8.
102. Black C, Clar C, Henderson R, et al. The clinical effectiveness of glucosamine and chondroitin supplements in slowing or arresting progression of osteoarthritis of the knee: a systematic review and economic evaluation. Health Technol Assess. 2009 Nov;13(52):1-148.
103. Gruenwald J, Petzold E, Busch R, Petzold HP, Graubaum HJ. Effect of glucosamine sulfate with or without omega-3 fatty acids in patients with osteoarthritis. Adv Ther. 2009 Sep;26(9):858-71.
104. Igarashi M, Kaga I, Takamori Y, Sakamoto K, Miyazawa K, Nagaoka I. Effects of glucosamine derivatives and uronic acids on the production of glycosaminoglycans by human synovial cells and chondrocytes. Int J Mol Med. 2011 Jun;27(6):821-7.
105. Igarashi M, Sakamoto K, Nagaoka I. Effect of glucosamine, a therapeutic agent for osteoarthritis, on osteoblastic cell differentiation. Int J Mol Med. 2011 Sep;28(3):373-9.
106. Ivanovska N, Dimitrova P. Bone resorption and remodeling in murine collagenase-induced osteoarthritis after administration of glucosamine. Arthritis Res Ther. 2011 Mar 16;13(2):R44.
107. Ng NT, Heesch KC, Brown WJ. Efficacy of a progressive walking program and glucosamine sulphate supplementation on osteoarthritic symptoms of the hip and knee: a feasibility trial. Arthritis Res Ther. 2010;12(1):R25.
108. Petersen SG, Saxne T, Heinegard D, et al. Glucosamine but not ibuprofen alters cartilage turnover in osteoarthritis patients in response to physical training. Osteoarthritis Cartilage. 2010 Jan;18(1):34-40.
109. Taccone-Gallucci M, Manca-di-Villahermosa S, Battistini L, et al. N-3 PUFAs reduce oxidative stress in ESRD patients on maintenance HD by inhibiting 5-lipoxygenase activity. Kidney Int. 2006 Apr;69(8):1450-4.
110. Calder PC. N-3 polyunsaturated fatty acids and inflammation: from molecular biology to the clinic. Lipids. 2003 Apr;38(4):343-52.
111. Aggarwal BB, Sung B. Pharmacological basis for the role of curcumin in chronic diseases: an age-old spice with modern targets. Trends Pharmacol Sci. 2009 Feb;30(2):85-94.
112. Safayhi H, Rall B, Sailer ER, Ammon HP. Inhibition by boswellic acids of human leukocyte elastase. J Pharmacol Exp Ther. 1997 Apr;281(1):460-3.
113. Safayhi H, Sailer ER, Ammon HP. Mechanism of 5-lipoxygenase inhibition by acetyl-11-keto-beta-boswellic acid. Mol Pharmacol. 1995 Jun;47(6):1212-6.